Funding
Medical Research Council (2017-present)
Academy of Medical Sciences (2014-2016 )
University of Oxford Medical Research Fund (2014-2017 )
National Institute of Health Research (2012-2016 )
Wellcome Trust (2007-2010)
Training and Qualifications
BSc (Hons) - University of Manchester (2002)
MBChB - University of Manchester (2003)
MRCP (UK) - Royal College of Physicians, London (2006)
PhD - University of Glasgow (2011)
MRCP (Neuro) - Royal College of Physicians and Surgeons of Glasgow (2011)
CCT (Neurology) - Joint Royal Colleges of Physicians Training Board (2015)
Biography
Simon Rinaldi was a medical undergraduate and postgraduate general medical trainee in Manchester. He moved to the University of Glasgow in 2007 to undertake a PhD looking into the neuroimmunology of inflammatory peripheral nerve disease, supervised by Professor Hugh Willison. Following 2 years of clinical neurology training in the East of Scotland, he was appointed as an NIHR Clinical Lecturer in Neurology at the University of Oxford in March 2012. During his lectureship he developed models of immune mediated axonal injury and demyelination using human induced pluripotent stem cells. He has now been awarded an MRC Clinician Scientist Fellowship to identify the specific antibodies responsible for the acute and chronic inflammatory neuropathies and to delineate their pathogenic mechanisms.
Research groups
Colleges
Websites
-
Mark Wallace
Collaborator
-
Hugh Willison
Collaborator
Collaborators
-
David Bennett
Professor of Neurology and Neurobiology
Simon Rinaldi
MRCP(Neuro), PhD
Professor of Neurology and Honorary Consultant Neurologist
- Clinical Director, Oxford Autoimmune Neurology Diagnostic Laboratory
Human cell-based assays to study the inflammatory neuropathies
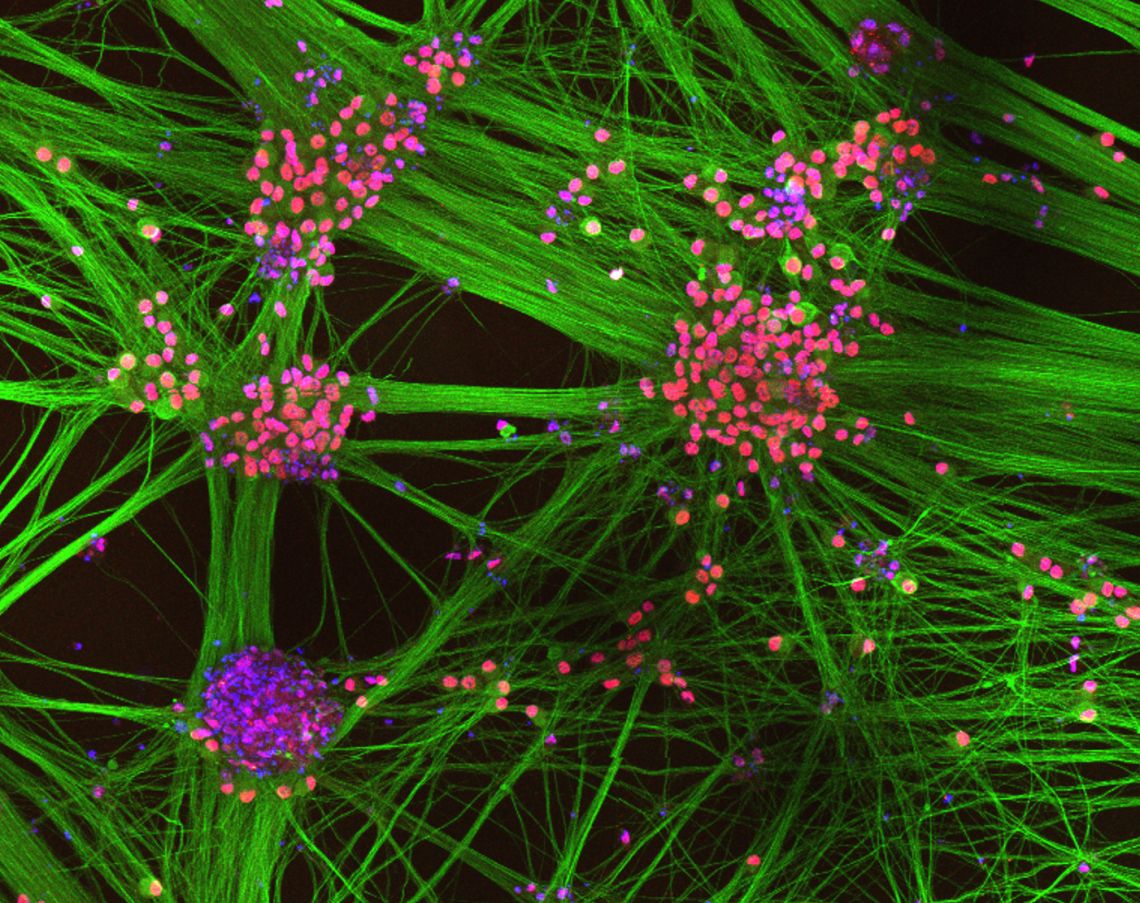
Sensory neurons derived from human stem cells
Research Summary
I lead the University of Oxford's programme of research into the inflammatory neuropathies.
Guillain-Barré syndrome (GBS) is a devastating, acute inflammatory neuropathy with substantial mortality as well as morbidity. There is an urgent need to improve upon the current situation where 4-5% of GBS patients die, 20-25% require mechanical ventilation, 14-20% have severe disability at 1 year, and many more are left with residual weakness, persistent pain and/or are unable to resume their former occupation.
Chronic inflammatory demyelinating polyradiculo-neuropathy (CIDP) and multifocal motor neuropathy (MMN) are chronic inflammatory neuropathies which produce significant long-term and often progressive disability with a sizeable economic cost.
Despite this, disease modifying therapies for these conditions have not advanced in over 20 years, the pathological mechanisms driving them are incompletely characterised, and there is no reliable way to identify early those patients who respond poorly to standard treatments and are thus most likely to benefit from additional therapy.
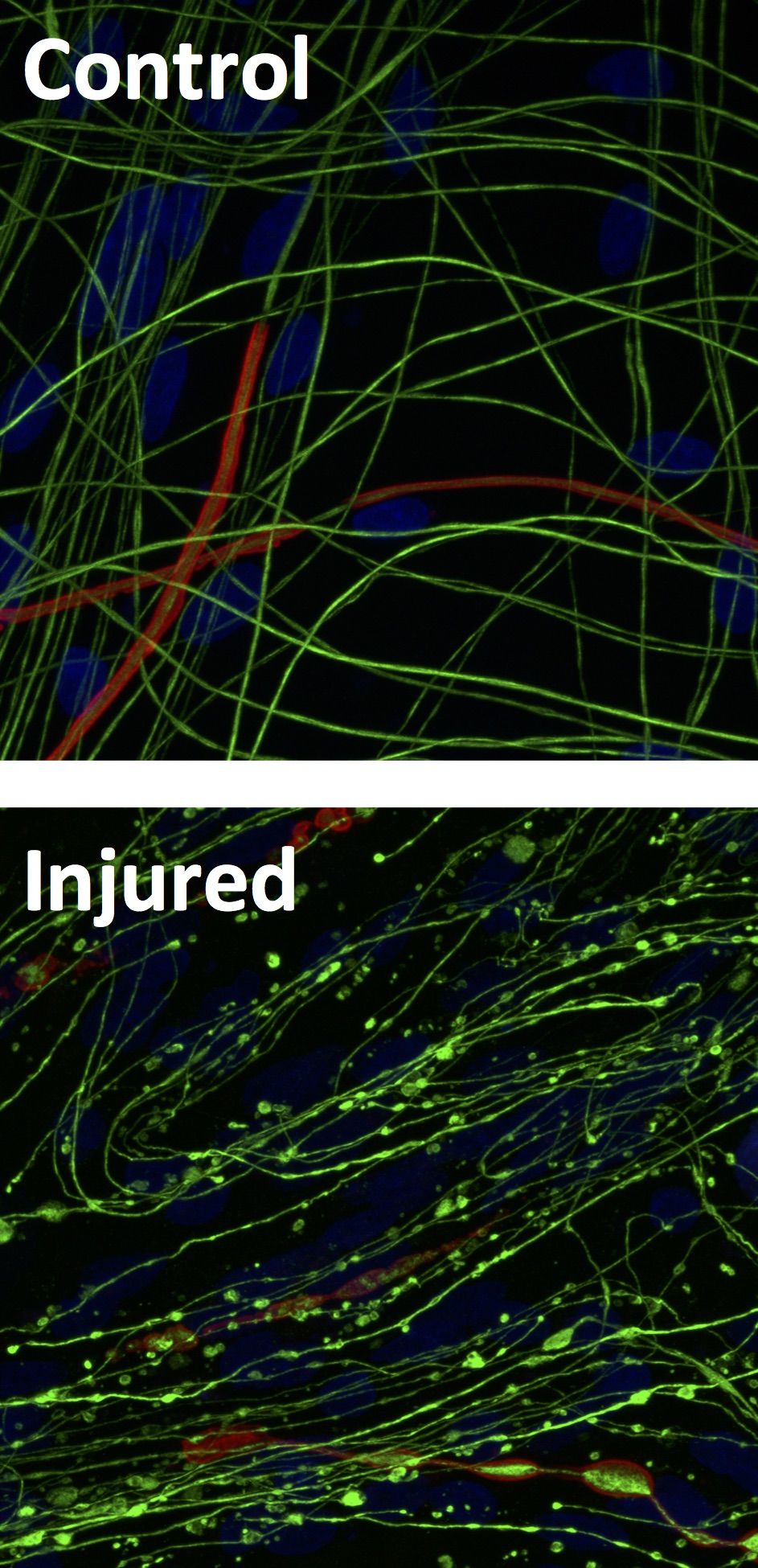
We have now successfully achieved myelination with human induced pluripotent stem cell derived neurons and Schwann cells in co-culture, a goal of numerous peripheral nerve laboratories around the world. Given the differences between rodent and human myelin, this offers clear advantages for the study of peripheral nerve injury and repair processes relevant to human health and disease.
Furthermore, the complex landscape of the live neural membrane, with interactions between neighbouring molecules, including those at specialised regions such as the node of Ranvier, is known to influence antibody-antigen interactions, yet cannot be accurately recapitulated in solid phase assays or in frozen sections. As well as better assessing these interactions, this technique allows the simultaneous evaluation of pathogenic effects and injury mechanisms.
Clinical Links
Our research also includes a clinical / observational study of chronic inflammatory neuropathy (Bio-SPiN) and the group contributes to and benefits from close links with the comprehensive and high-quality clinical-serological database of over 1400 patients encapsulated in the International GBS Outcome Study (IGOS). We have previously been involved in therapeutic trials of fingolimod for CIDP and eculizumab for GBS.
Key publications
Natural Killer Cells Degenerate Intact Sensory Afferents following Nerve Injury
Journal article
Davies AJ. et al, (2019), Cell, 176, 716 - 728.e18
Co-cultures with stem cell-derived human sensory neurons reveal regulators of peripheral myelination
Journal article
Clark AJ. et al, (2017), Brain, 140, 898 - 913
Antibodies to Heteromeric Glycolipid Complexes in Guillain-Barré Syndrome
Journal article
Rinaldi S. et al, (2013), PLoS ONE, 8, e82337 - e82337
The neuropathic potential of anti-GM1 autoantibodies is regulated by the local glycolipid environment in mice
Journal article
Greenshields KN. et al, (2009), Journal of Clinical Investigation, 119, 595 - 610
Recent publications
Clinical Features and Management of Optic Neuritis in a Cohort of Patients with Combined Central and Peripheral Demyelination
Journal article
Moodley K. et al, (2026), Neuro-Ophthalmology, 50, 143 - 158
Measurement properties of the Inflammatory Rasch-built Overall Disability Scale (I-RODS) in patients with Guillain–Barré syndrome
Journal article
Pelouto F. et al, (2026), Journal of Neurology, 273
NKG2D receptor ligands are cell surface biomarkers for injured murine and human nociceptive sensory neurons
Journal article
Wang S. et al, (2025), Journal of Neuroinflammation, 23
Plasma periaxin is a biomarker of peripheral nerve demyelination
Journal article
Bellanti R. et al, (2025), Brain, 148, 4448 - 4460
Chronic Inflammatory Demyelinating Polyradiculoneuropathy ( CIDP ) Developing During Tacrolimus Treatment: A Case Series
Journal article
Evans MC. et al, (2025), Muscle & Nerve, 72, 1289 - 1293
Transfected Cell Based Immunoassays for Nodal / Paranodal Antibodies
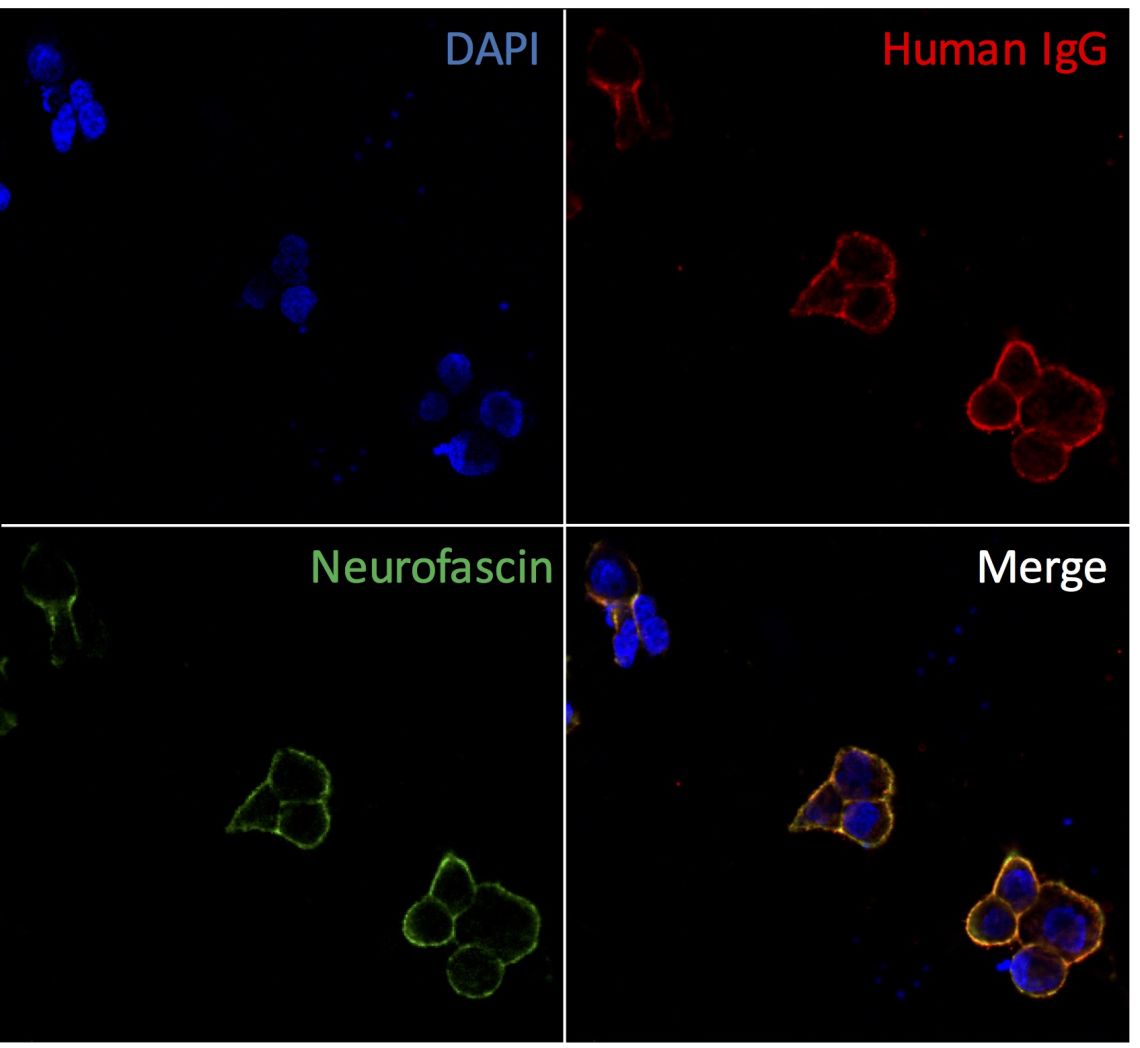
In parallel to to work using human induced pluripotent stem cells, we have also established a transfected cell based assay to detect antibodies against the nodal and paranodal proteins neurofascin-155, neurofascin-140/186, contactin-1 and Caspr1, known to be associated with certain subtypes of CIDP and combined central and peripheral demyelination (CCPD). These are now available for clinical testing.
The request form, which should be emailed to us in parallel ago any samples being sent, is available for download below.
Please note, the request from and instructions for sending these samples are now available via the OUH NHS Immunology website.
- Request form for nodal / paranodal antibody testing
- Microsoft Office - Word Document 498.2 KB
Please ensure this is completed electronically, saved as a word document, and sent at the same time as the sample.
- Instructions for sending samples for paranodal antibody testing
- PDF document 134.8 KB
Instructions for sending samples for paranodal antibody testing
Immune mediated axonal injury